 Astellas will conduct all subsequent research, development, manufacturing, and commercialization activities. 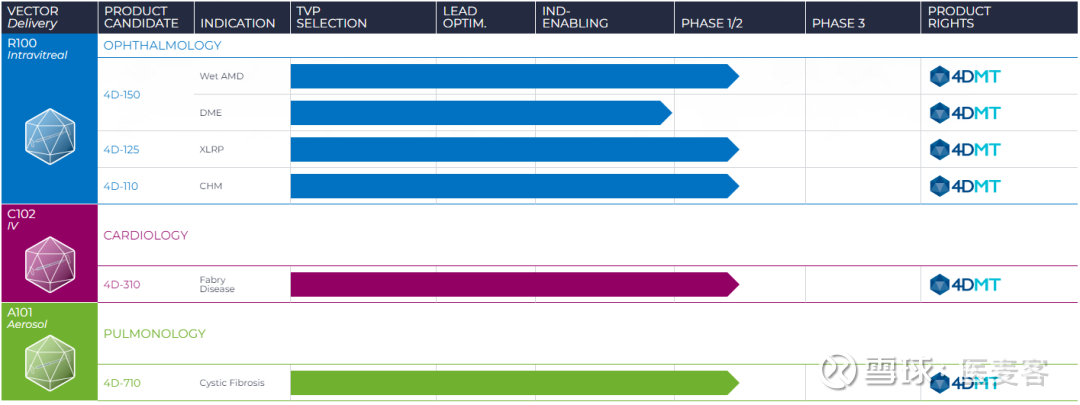
Under the terms of the agreement, 4DMT will provide its proprietary R100 vector technology to Astellas to deliver Astellas' unique genetic payloads for the treatment of rare monogenic diseases. All three 4DMT clinical-stage ophthalmic product candidates utilize the R100 vector, including 4D-150 for wet age-related macular degeneration and diabetic macular edema. It has the ability to penetrate the internal limiting membrane barrier and to efficiently transduce the entire retina, resulting in robust transgene expression within retinal cells. R100 is an adeno-associated virus (AAV) vector invented by 4DMT for intravitreal delivery. (NASDAQ: FDMT) (CEO: David Kirn, MD, "4DMT") today announced a license agreement under which Astellas gains rights to utilize the intravitreal retinotropic R100* vector invented by 4DMT for one genetic target implicated in rare monogenic ophthalmic disease(s), with options to add up to two additional targets implicated in rare monogenic ophthalmic diseases after paying additional option exercise fees. (TSE: 4503) (President and CEO: Naoki Okamura, "Astellas") and 4D Molecular Therapeutics, Inc. No representation is made as to the safety or effectiveness of 4D-150, 4D-710, 4D-310, 4D-125, or 4D-110 for the therapeutic uses for which they are being studied.ĤD Molecular Therapeutics™, 4DMT™, Therapeutic Vector Evolution™, and the 4DMT logo are trademarks of 4DMT.TOKYO and EMERYVILLE, Calif., J/PRNewswire/ - Astellas Pharma Inc. The 4D preclinical product candidates in development are: 4D-175 for geographic atrophy and 4D-725 for AATLD.ĤD-150, 4D-710, 4D-310, 4D-125, and 4D-110 are our product candidates in clinical development and have not yet been approved for marketing by the US FDA or any other regulatory authority. 4DMT is currently advancing five product candidates in clinical development: 4D-150 for wet AMD and DME, 4D-710 for cystic fibrosis lung disease, 4D-310 for Fabry disease cardiomyopathy, 4D-125 for XLRP, and 4D-110 for choroideremia. The 4DMT customized and evolved vectors were invented with the goal of being delivered at relatively low doses through clinically routine, well-tolerated, and minimally invasive routes of administration, transducing diseased cells in target tissues efficiently, having reduced immunogenicity and, where relevant, having resistance to pre-existing antibodies. The Company is initially focused on five clinical-stage product candidates in three therapeutic areas for both rare and large market diseases: ophthalmology, pulmonology, and cardiology. All of our vectors are proprietary to 4DMT and were invented at 4DMT, including the vectors utilized in our clinical-stage and preclinical pipeline product candidates: R100, A101, and C102. 4DMT seeks to unlock the full potential of genetic medicines using its proprietary invention platform, Therapeutic Vector Evolution, which combines the power of the Nobel Prize-winning technology, directed evolution, with approximately one billion synthetic AAV capsid-derived sequences to invent customized and evolved vectors for use in our product candidates. 
ĤDMT is a clinical-stage biotherapeutics company harnessing the power of directed evolution for genetic medicines targeting large market diseases. Goldman Sachs Global Healthcare Conference Fireside Chat Date:Īrchived copies of the webcasts will be available for up to one year by visiting the “Investors” section of the 4DMT website at. Jefferies Global Healthcare Conference Fireside Chat Date: (Nasdaq: FDMT), a clinical-stage biotherapeutics company harnessing the power of directed evolution for genetic medicines targeting large market diseases, announced today it will participate in the following upcoming investor conferences: EMERYVILLE, Calif., J(GLOBE NEWSWIRE) - 4D Molecular Therapeutics, Inc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
